Blog > Emerging Trends > Genetic Testing in Psychiatry | Pharmacogenomic Testing Guide
Genetic Testing in Psychiatry: How DNA Testing Guides Mental Health Medication Choices
Genetic testing in psychiatry is transforming how clinicians select and manage mental health medications. By analyzing a patient’s DNA, psychiatric pharmacogenomic testing can reveal how medications are metabolized, predict treatment response, and identify potential side effects before they occur. This guide explains how DNA testing for mental health medications works, when it’s most useful, how accurate it is, and whether genetic testing for antidepressants is covered by insurance — helping clinicians and patients make more informed, individualized treatment decisions.

Last Updated: March 27, 2026


What You'll Learn
- How genetic testing in psychiatry guides medication selection and dosing
- What a psychotropic test reveals about metabolism, response, and side effects
- When pharmacogenomic testing is most useful in clinical practice
- How accurate genetic testing for psychiatric medications really is
- Whether genetic testing for antidepressants is covered by insurance
- How clinicians can document medical necessity to improve coverage approval
Contents
-
How DNA Testing for Mental Health Medications Guides Treatment Decisions
-
When Is Genetic Testing for Psychiatric Medications a Good Idea?
- How Accurate is Genetic Testing for Psychiatric Medications?
- Is Genetic Testing for Antidepressants Covered by Insurance?
- Ethical Considerations for Clinicians
- Navigating Precision Psychiatry with Genetic Insights Using ICANotes
- FAQ: Genetic Testing for Psychiatric Medication
Finding the right psychiatric medication often means months of trial and error — cycling through drugs that don't work, managing side effects that shouldn't have to be endured, and waiting for relief that feels perpetually out of reach. But advances in genetic testing in psychiatry are changing that equation. By analyzing a patient's DNA, clinicians can now make more informed, individualized medication decisions from the start.
This approach, known as psychiatric pharmacogenomic testing, examines specific genes tied to drug metabolism, neurotransmitter pathways, and receptor interactions. The result is a pharmacogenetic profile that helps mental health professionals move beyond one-size-fits-all prescribing toward true precision medicine.
In this guide, we'll break down how DNA testing for mental health medications works, what it reveals, when it's most useful, how accurate it is, and whether insurance will cover it.
What is Psychiatric Pharmacogenomic Testing?
Psychiatric pharmacogenomic testing is a type of genetic test that identifies specific genetic markers — known as alleles — linked to how an individual metabolizes medications, responds to neurotransmitter activity, and tolerates different drug classes. These markers are most commonly associated with drug metabolism enzymes (such as the cytochrome P450 family), neurotransmitter receptors, and cellular signaling pathways.
Together, these markers form a patient's pharmacogenetic profile: a biological blueprint that helps clinicians predict which medications are most likely to work, which may cause adverse reactions, and what dosages are appropriate.
How Psychiatric Pharmacogenomic Testing Works
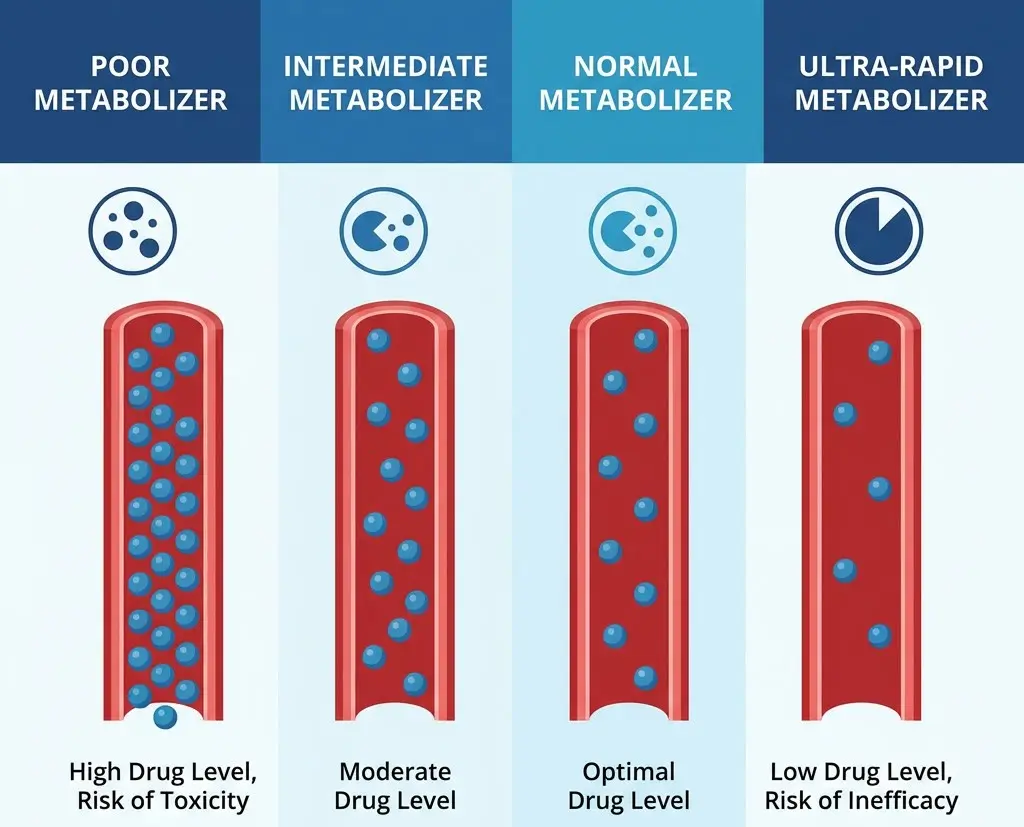
A psychotropic test typically begins with a simple cheek swab or blood draw. The sample is sent to a laboratory, where technicians analyze a targeted panel of genes. The resulting report categorizes the patient by metabolizer status for various drug-processing enzymes — for example, identifying someone as a poor metabolizer, intermediate metabolizer, normal metabolizer, or ultra-rapid metabolizer of a given enzyme.
Clinicians then interpret these results alongside the patient's clinical history, current symptoms, and treatment goals. Genetic testing doesn't replace clinical judgment — it sharpens it. The data gives mental health professionals a more complete picture, helping them select medications and calibrate dosages with greater confidence.
In many reports, medications are grouped into categories based on how well they align with the patient’s genetic profile, often using color-coded guidance to indicate potential concerns or compatibility. This allows clinicians to quickly identify which options may require dose
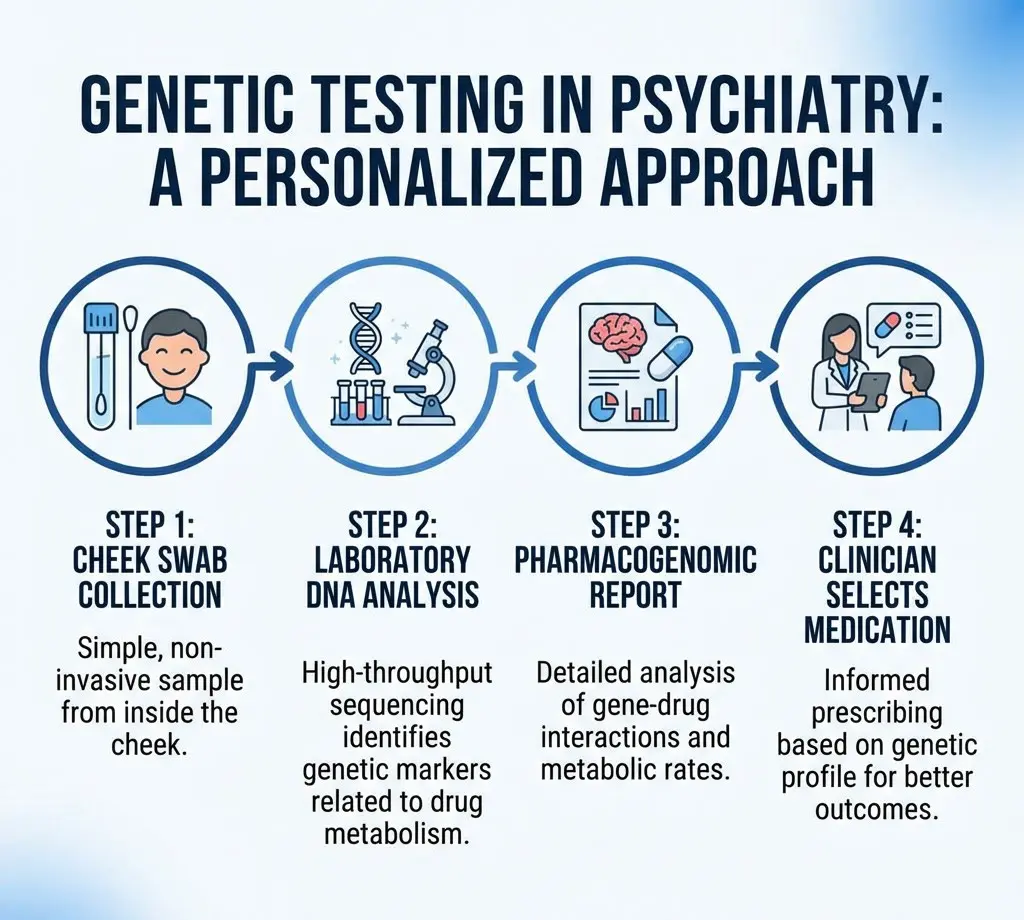
adjustments, closer monitoring, or avoidance altogether. When combined with a thorough assessment, these insights support more precise, individualized prescribing and can significantly reduce the time it takes to find an effective treatment.
What a Psychotropic Test Reveals
A psychotropic test uncovers several categories of clinically useful information:
Metabolism Rates
Perhaps the most actionable insight is how quickly or slowly a patient's body processes specific medications. A poor metabolizer may experience heightened side effects at standard doses, while an ultra-rapid metabolizer may clear a drug too quickly for it to be effective.
Medication Responsiveness
Genetic markers linked to neurotransmitter receptors and signaling pathways can indicate whether a patient is more likely to respond to certain drug classes — for instance, whether an SSRI or SNRI is a better starting point for depression treatment.
Side Effect Predisposition
Some genetic variants are associated with a higher risk of specific adverse reactions. Identifying these vulnerabilities in advance allows clinicians to steer away from medications that could worsen a patient's condition.
Drug-Drug Interaction Risk
For patients taking multiple medications — common in cases of dual diagnosis or complex comorbidities — genetic testing can highlight potential pharmacokinetic interactions that might otherwise be difficult to anticipate.
How Genetic Testing in Psychiatry Works
The integration of psychiatry genetic testing could herald a new era of precision medicine in psychiatry.
The foundation of genetic testing for psychiatric medication rests on identifying specific genetic markers, known as alleles, that are key to treatment success. These markers, often linked to drug metabolism enzymes, neurotransmitter receptors and cellular signaling pathways, provide a glimpse into the patient's unique pharmacogenetic profile.
Clinicians must decode the complex interplay between genetic markers and treatment outcomes, making sense of the probabilities and tendencies that genetic information provides. With this knowledge, mental health professionals can tailor their treatment strategies, adjusting medication dosages and selecting drugs that align with a patient's genetic predispositions.
How DNA Testing for Mental Health Medications Guides Treatment Decisions
The clinical value of DNA testing for mental health medications is most apparent in the decisions it enables: faster identification of effective drugs, fewer unnecessary side effects, and more strategic treatment planning overall.
Reducing Trial and Error with Psychotropic Testing
The traditional approach to psychiatric medication selection involves starting a drug, waiting weeks to assess its effects, adjusting or switching if it fails, and repeating the cycle. For many patients, this process stretches across months or even years.
Psychotropic testing compresses that timeline. Rather than prescribing based on population-level averages and waiting to see what happens, clinicians can use a patient's genetic data to narrow the field of likely effective medications from the outset. This doesn't eliminate clinical judgment or monitoring, but it gives the treatment process a more informed starting point — reducing the number of failed trials and the cumulative burden they place on patients.
Want to see how this works in a real clinical scenario? Watch our on-demand webinar, where we compare pharmacogenomic test results from two different companies for the same patient.
Faster Antidepressant Selection Through Genetic Testing
Depression treatment is one of the areas where genetic testing in psychiatry has the most immediate practical impact. When a patient presents with major depressive disorder, clinicians often face a choice between multiple drug classes and dozens of individual medications, each with different efficacy and side-effect profiles.
Genetic markers can guide this decision. For example, a patient's metabolizer status for CYP2D6 and CYP2C19 — two enzymes heavily involved in processing SSRIs and SNRIs — can indicate which antidepressants are most likely to reach therapeutic levels without causing excessive side effects. This genetic insight helps clinicians prescribe more precisely, getting patients to relief faster.
Predicting Side Effects with Pharmacogenomic Testing
One of the more powerful applications of pharmacogenomic testing is its ability to anticipate adverse reactions before they happen. Certain genetic variants are well-documented predictors of side effects like weight gain, sedation, QT prolongation, or serotonin syndrome risk.
By identifying these vulnerabilities in advance, clinicians can proactively avoid problematic medications rather than discovering the problem after a patient has already experienced the side effect. This is especially valuable for patients who have a history of poor medication tolerance or who are taking other drugs that compound risk.
Tailoring Dual Diagnosis Strategies
For patients managing co-occurring conditions — such as depression alongside a substance use disorder, or anxiety combined with ADHD — treatment planning becomes significantly more complex. Each condition may call for medications that interact with one another, and the genetic factors influencing one condition's treatment may affect the other.
Genetic testing helps untangle this complexity. By mapping a patient's pharmacogenetic profile across multiple drug pathways, clinicians can identify treatment combinations that are more likely to be safe and effective in tandem, rather than optimizing for one condition at the expense of another.
When is Genetic Testing in Psychiatry a Good Idea?
Genetic testing can add value across a broad range of clinical scenarios, but there are specific situations where it becomes an especially powerful tool.
Genetic Testing for Treatment-Resistant Conditions
When a patient has tried multiple medications without adequate response, genetic testing offers a potential path forward. Treatment resistance is often a signal that something in the patient's biology is interfering with standard pharmacological approaches — and genetic markers can help identify what that something is.
For patients who have cycled through several failed medication trials, a psychotropic test can reveal whether they are poor or ultra-rapid metabolizers of the drugs they've been prescribed, or whether genetic factors make them more likely to respond to an entirely different drug class. This information can break a frustrating cycle of ineffective treatment and open up new avenues for relief.
Which Psychiatric Conditions Benefit from DNA Testing?
While psychiatric pharmacogenomic testing is relevant across many diagnoses, it tends to deliver the most value in conditions characterized by complexity and variability in treatment response.
Bipolar disorder, with its interplay of mood states and the need to balance mood stabilizers, antipsychotics, and sometimes antidepressants, is a prime example. Genetic insights can help clinicians navigate the complexity of multi-drug regimens and reduce the risk of triggering mood episodes with the wrong medication choice.
Major depressive disorder, particularly cases that haven't responded to first-line treatments, is another area where genetic testing frequently proves its worth. The same is true for anxiety disorders, ADHD, and PTSD — conditions where medication selection often involves significant guesswork without genetic data.
Cases involving dual diagnoses or complex comorbidities also benefit disproportionately, as genetic testing can illuminate interactions between treatments for different conditions that might otherwise go unnoticed.
Pediatric Psychiatry
Children and adolescents present distinct treatment challenges. Their bodies are still developing, their physiology differs from that of adults, and they may metabolize medications at different rates. Genetic testing is particularly valuable in pediatric psychiatry because it can help clinicians avoid unnecessary medication trials during a critical developmental window.
For young patients, the stakes of getting medication wrong are high — not only in terms of side effects, but also in terms of the psychological impact of prolonged untreated or inadequately treated symptoms. DNA testing for mental health medications provides a data point that can accelerate the path to effective treatment and reduce the burden on both the child and their family.
Individualizing Geriatric Care
Older adults face their own set of pharmacological challenges. Age-related changes in liver function, kidney clearance, and body composition all affect how medications are processed. Polypharmacy — the use of multiple medications simultaneously — is also more common in this population, increasing the risk of drug interactions.
Genetic testing adds a critical layer of information to geriatric treatment planning. By understanding a patient's metabolizer status, clinicians can adjust dosages to account for both genetic and age-related factors, minimizing the risk of adverse reactions and improving the likelihood of a positive treatment outcome.
How Accurate is Genetic Testing for Psychiatric Medications?
This is one of the most common questions patients and clinicians ask, and the answer is nuanced.
Genetic testing for psychiatric medications is reliable at what it directly measures: identifying genetic variants and categorizing metabolizer status. When a test reports that a patient is a poor metabolizer of CYP2D6, for example, that finding is highly accurate. The laboratory science behind identifying these alleles is well-established and reproducible.
Where the picture becomes more complex is in translating those genetic findings into clinical predictions. Medication response is multifactorial — genetics plays a significant role, but so do environmental factors, co-occurring conditions, diet, other medications, and the inherent variability of psychiatric conditions themselves. A genetic test can tell you that a patient is likely to process a certain SSRI slowly, but it cannot guarantee that a different SSRI will resolve their depression.
That said, the evidence supporting pharmacogenomic-guided prescribing is growing. Several studies have found that patients whose treatment was informed by genetic testing experienced better outcomes, including higher response and remission rates, compared to those who received treatment as usual. The Clinical Pharmacogenetics Implementation Consortium (CPIC) publishes evidence-graded guidelines for specific gene-drug pairs, and the strength of evidence varies — it's strongest for well-studied interactions like CYP2D6 and CYP2C19 with SSRIs, tricyclic antidepressants, and certain antipsychotics.
The most accurate way to think about genetic testing is as a clinical decision-support tool. It doesn't provide certainty, but it significantly improves the probability of making a good medication choice on the first or second attempt rather than the fourth or fifth. Its accuracy is highest when the results are interpreted by a clinician who integrates them with a comprehensive clinical evaluation — not as a standalone oracle, but as one powerful input in a broader treatment picture.
Is Genetic Testing for Antidepressants Covered by Insurance?
Coverage for genetic testing for antidepressants has expanded significantly in recent years, though it still varies based on your insurance provider, plan, and clinical circumstances.
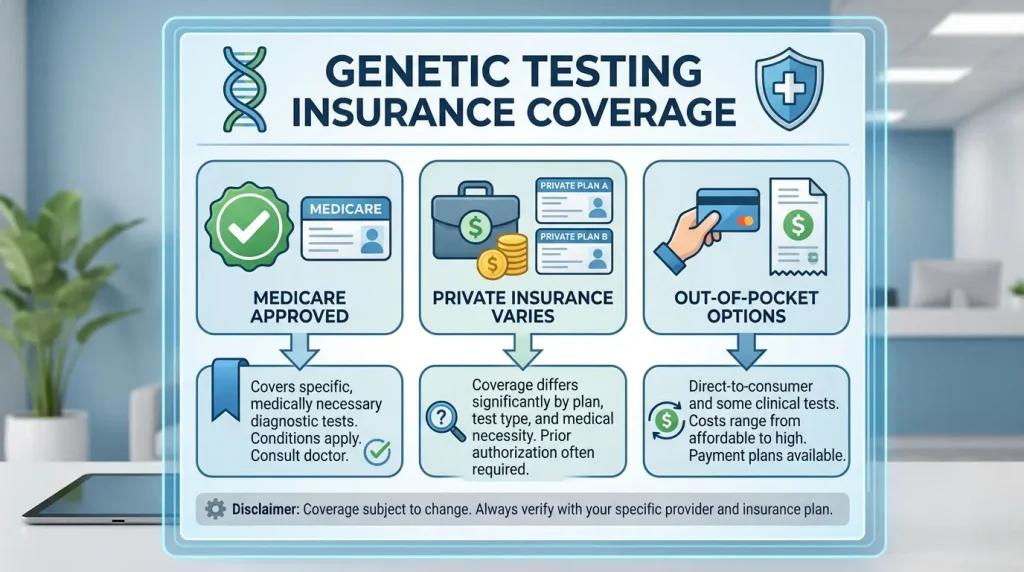
Medicare Coverage
Medicare has provided coverage for pharmacogenomic testing for antidepressants in many cases, particularly when a patient has a documented history of treatment failure or adverse drug reactions. Coverage typically requires that the test be ordered by a treating physician and that there is a clear clinical rationale — such as a patient who has tried and failed at least one antidepressant.
Private Insurance Coverage
Private insurance coverage varies more widely. Many major insurers now cover pharmacogenomic testing when medical necessity is documented, but the specific criteria differ from plan to plan. Some insurers require prior authorization, while others will cover testing only after a certain number of failed medication trials. It’s always advisable for patients to contact their insurance provider directly to confirm coverage before testing.
Out-of-Pocket Options
For patients without coverage, many pharmacogenomic testing companies offer financial assistance programs, sliding-scale pricing, or cash-pay options that can reduce out-of-pocket costs significantly. Some companies cap patient costs at a few hundred dollars regardless of insurance status.
How Clinicians Can Support Coverage
Thorough documentation of medical necessity is the single most important factor in securing insurance approval. This includes recording the patient’s medication history, documenting failed trials and adverse reactions, and clearly articulating why genetic testing is clinically indicated. A well-documented case makes it significantly easier for insurers to approve the test.
If you're a patient considering pharmacogenomic testing, ask your prescribing clinician about your options and contact your insurer to understand your plan's specific requirements. If you're a clinician, building pharmacogenomic test orders into your clinical workflow — with proper documentation — can help ensure your patients have access to this tool when they need it.
Ethical Considerations for Clinicians
As genetic testing in psychiatry becomes more widespread, mental health professionals carry a responsibility to navigate its ethical dimensions thoughtfully.
Privacy and Data Security
Genetic information is inherently personal. Clinicians must ensure that pharmacogenomic data is stored, transmitted, and handled with the same rigor as any other sensitive health information — and in many cases, with additional safeguards. Patients should understand how their genetic data will be used, who will have access to it, and how long it will be retained.
The Art of Interpretation
A psychotropic test is not a crystal ball. It produces a mosaic of probabilities that requires skilled clinical interpretation. Genetic insights must be weighed alongside clinical evaluations, patient history, and the full context of a patient's mental health picture. Over-reliance on genetic data without adequate clinical integration risks reducing a complex human being to a set of alleles — a failure of both science and care.
Informed Patient Collaboration
Patients deserve a clear understanding of what genetic testing can and cannot do. Engaging in shared decision-making means explaining the benefits, limitations, and implications of testing before it's ordered — not after. Mental health professionals should foster open dialogue, ensuring patients understand that genetic insights are one valuable input in a collaborative treatment process, not a unilateral prescription generator.
Cultural and Social Sensitivity
Genetic testing does not exist in a vacuum. Cultural beliefs, historical mistrust of medical research in certain communities, and differing attitudes toward genetic information all influence how patients perceive and engage with pharmacogenomic testing. Clinicians must approach testing recommendations with cultural humility, respecting diverse perspectives and ensuring that a patient's values and beliefs are part of the conversation.
Navigating Precision Psychiatry with Genetic Insights Using ICANotes
The integration of genetic testing in psychiatry represents a meaningful step toward precision behavioral health — treatment strategies built around the individual rather than the average. From psychiatric pharmacogenomic testing that reveals how a patient metabolizes medication, to DNA testing for mental health medications that predicts side effects and drug interactions, these tools give clinicians a sharper, more informed foundation for treatment decisions.
At ICANotes, we're committed to supporting mental health professionals as these tools become a standard part of clinical practice. Our platform is designed to help you integrate genetic insights into your treatment planning, documentation, and workflow — so you can spend less time on administrative burden and more time delivering the personalized care your patients deserve.
Start Your 30-Day Free Trial
Experience the most intuitive, clinically robust EHR designed for behavioral health professionals, built to streamline documentation, improve compliance, and enhance patient care.
- Complete Notes in Minutes - Purpose-built for behavioral health charting
- Always Audit-Ready – Structured documentation that meets payer requirements
- Keep Your Schedule Full – Automated reminders reduce costly no-shows
- Engage Clients Seamlessly – Secure portal for forms, messages, and payments
- HIPAA-Compliant Telehealth built into your workflow

Frequently Asked Questions: Genetic Testing Psychiatry
Related Posts
About the Author
Dr. October Boyles is a behavioral health expert and clinical leader with extensive expertise in nursing, compliance, and healthcare operations. With a Doctor of Nursing Practice (DNP) and advanced degrees in nursing, she specializes in evidence-based practices, EHR optimization, and improving outcomes in behavioral health settings. Dr. Boyles is passionate about empowering clinicians with the tools and strategies needed to deliver high-quality, patient-centered care.









